Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
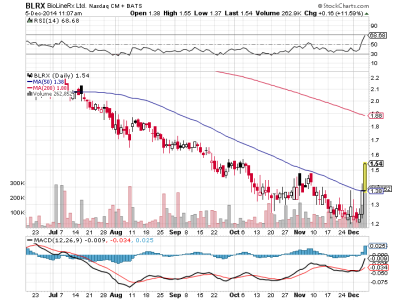
blrx
- Creatore Discussione majorino
- Data di inizio
-
 Due nuove obbligazioni Societe Generale, in Euro e in Dollaro USA
Due nuove obbligazioni Societe Generale, in Euro e in Dollaro USA

Societe Generale porta sul segmento Bond-X (EuroTLX) di Borsa Italiana due obbligazioni, una in EUR e una in USD, a tasso fisso decrescente con durata massima di 15 anni e possibilità di rimborso anticipato annuale a discrezione dell’Emittente.
Per continuare a leggere visita questo LINK
Più opzioni
Chi ha risposto?puo' essere un buon investimento per l'anno appena iniziato
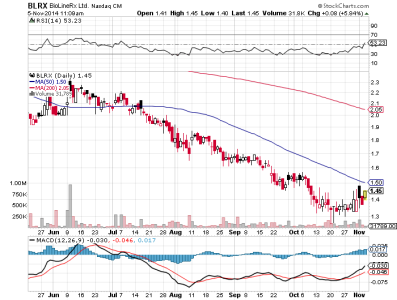
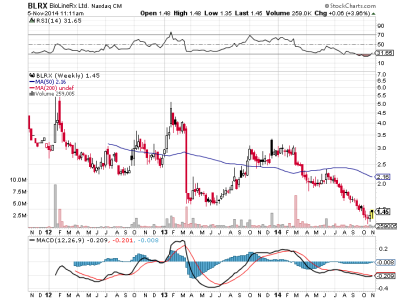
last 3,37

4,34
LOROCIU65
""ONLY THE BRAVE""
- Registrato
- 1/3/06
- Messaggi
- 31.837
- Punti reazioni
- 689
non la conosco..da approfondire..AFTER +43%
US Patent and Trademark Office (USPTO) ha recentemente pubblicato un nuovo brevetto degli Stati Uniti per BioLineRx, Ltd. 's ( BLRX ) BL-8040 (brevetto n ° 8.455.450).
Il brevetto, che scadrà nel novembre 2029, coprirà BL-8040 per l'utilizzo di sua composizione per l'ottenimento di cellule precursori emopoietici (ad esempio cellule staminali) per l'uso nel trapianto di midollo osseo.
Questo nuovo brevetto aumenta il portafoglio di brevetti esistente di BL-8040, che ha 6 brevetti rilasciati e 29 domande di brevetti per nuove indicazioni in tutto il mondo.
BioLineRx 'BL-8040 è in uno studio di fase II per il trattamento della leucemia mieloide acuta (AML). Lo studio multicentrico, in aperto, di fase II sta valutando la sicurezza e l'efficacia di dosi crescenti ripetute di BL-8040 in adulti affetti da recidiva / refrattaria leucemia mieloide acuta.
Gli endpoint primari includono la sicurezza e la tollerabilità di BL-8040. Gli endpoint secondari includono il profilo farmacocinetico del candidato e di una valutazione di efficacia.
Il mese scorso, BioLineRx annunciato che BL-8040 ha mostrato i risultati pre-clinici positivi per il trattamento della trombocitopenia (riduzione della produzione di piastrine). I dati sono stati pubblicati sul British Journal of Hematology.
L'effetto di dosi ripetute di BL-8040 in buona salute così come topi trombocitopenici indotta da chemioterapia è stato valutato in uno studio pre-clinico.
Il numero di megacariociti nel midollo osseo migliorato significativamente con la somministrazione ripetuta di BL-8040. Megacariociti sono le cellule che producono le piastrine.
Inoltre, BL-8040 ha aumentato il numero di cellule progenitrici ematopoietiche nel midollo osseo e nel sangue. Queste cellule producono le piastrine così come globuli rossi e bianchi. La gravità e la durata della trombocitopenia indotta da chemioterapia e citopenia sono diminuiti in modo significativo.
Trombocitopenia è un effetto collaterale frequente della chemioterapia. Capacità del candidato di ridurre trombocitopenia rende più attraente per l'uso del trattamento del cancro del sangue.
Si ricorda agli investitori che, nel settembre 2012, i diritti per BL-8040 erano in licenza da BioLineRx da Biokine Therapeutics Ltd.
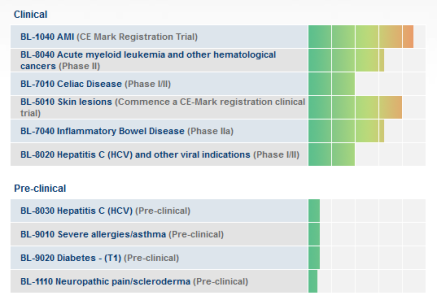
Alcuni dei candidati sviluppati da BioLineRx includono BL-5010 per il trattamento di lesioni cutanee, BL-7040 per il trattamento della malattia infiammatoria intestinale (IBD) e BL-1021 per il trattamento del dolore neuropatico.
In Apr 2013, BioLineRx annunciato incoraggianti risultati di uno studio di fase IIa su BL-7040. I risultati dello studio hanno rivelato che BL-7040 è sicuro ed efficace per il trattamento di pazienti affetti da colite ulcerosa, che è una forma di malattia infiammatoria intestinale.
BioLineRx porta un Zacks Classifica # 3 (attesa). Le aziende che attualmente sembrano ben posizionati includono Actelion Ltd. ( ALIOF ) e Gilead Sciences Inc. ( GILD - Analyst Report ) con un Rango Zacks # 1 (Strong Buy) e Jazz Pharmaceuticals ( JAZZ - Snapshot relazione ) con un Zacks # 2 (Buy).
US Patent and Trademark Office (USPTO) ha recentemente pubblicato un nuovo brevetto degli Stati Uniti per BioLineRx, Ltd. 's ( BLRX ) BL-8040 (brevetto n ° 8.455.450).
Il brevetto, che scadrà nel novembre 2029, coprirà BL-8040 per l'utilizzo di sua composizione per l'ottenimento di cellule precursori emopoietici (ad esempio cellule staminali) per l'uso nel trapianto di midollo osseo.
Questo nuovo brevetto aumenta il portafoglio di brevetti esistente di BL-8040, che ha 6 brevetti rilasciati e 29 domande di brevetti per nuove indicazioni in tutto il mondo.
BioLineRx 'BL-8040 è in uno studio di fase II per il trattamento della leucemia mieloide acuta (AML). Lo studio multicentrico, in aperto, di fase II sta valutando la sicurezza e l'efficacia di dosi crescenti ripetute di BL-8040 in adulti affetti da recidiva / refrattaria leucemia mieloide acuta.
Gli endpoint primari includono la sicurezza e la tollerabilità di BL-8040. Gli endpoint secondari includono il profilo farmacocinetico del candidato e di una valutazione di efficacia.
Il mese scorso, BioLineRx annunciato che BL-8040 ha mostrato i risultati pre-clinici positivi per il trattamento della trombocitopenia (riduzione della produzione di piastrine). I dati sono stati pubblicati sul British Journal of Hematology.
L'effetto di dosi ripetute di BL-8040 in buona salute così come topi trombocitopenici indotta da chemioterapia è stato valutato in uno studio pre-clinico.
Il numero di megacariociti nel midollo osseo migliorato significativamente con la somministrazione ripetuta di BL-8040. Megacariociti sono le cellule che producono le piastrine.
Inoltre, BL-8040 ha aumentato il numero di cellule progenitrici ematopoietiche nel midollo osseo e nel sangue. Queste cellule producono le piastrine così come globuli rossi e bianchi. La gravità e la durata della trombocitopenia indotta da chemioterapia e citopenia sono diminuiti in modo significativo.
Trombocitopenia è un effetto collaterale frequente della chemioterapia. Capacità del candidato di ridurre trombocitopenia rende più attraente per l'uso del trattamento del cancro del sangue.
Si ricorda agli investitori che, nel settembre 2012, i diritti per BL-8040 erano in licenza da BioLineRx da Biokine Therapeutics Ltd.
Alcuni dei candidati sviluppati da BioLineRx includono BL-5010 per il trattamento di lesioni cutanee, BL-7040 per il trattamento della malattia infiammatoria intestinale (IBD) e BL-1021 per il trattamento del dolore neuropatico.
In Apr 2013, BioLineRx annunciato incoraggianti risultati di uno studio di fase IIa su BL-7040. I risultati dello studio hanno rivelato che BL-7040 è sicuro ed efficace per il trattamento di pazienti affetti da colite ulcerosa, che è una forma di malattia infiammatoria intestinale.
BioLineRx porta un Zacks Classifica # 3 (attesa). Le aziende che attualmente sembrano ben posizionati includono Actelion Ltd. ( ALIOF ) e Gilead Sciences Inc. ( GILD - Analyst Report ) con un Rango Zacks # 1 (Strong Buy) e Jazz Pharmaceuticals ( JAZZ - Snapshot relazione ) con un Zacks # 2 (Buy).
Mallella
So di non sapere
- Registrato
- 11/5/01
- Messaggi
- 4.813
- Punti reazioni
- 105
Ottima pipeline e 30 milioni in cassa.
News di oggi: http://finance.yahoo.com/news/biolinerx-announces-successful-final-results-120000146.html
News di oggi: http://finance.yahoo.com/news/biolinerx-announces-successful-final-results-120000146.html
Allegati
Mallella
So di non sapere
- Registrato
- 11/5/01
- Messaggi
- 4.813
- Punti reazioni
- 105
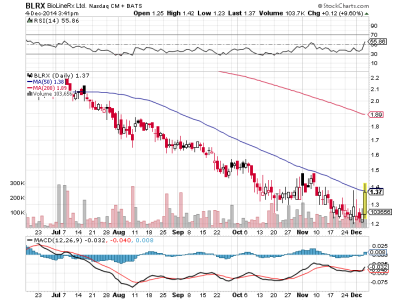
Trimestrale senza infamia e senza lode
http://finance.yahoo.com/news/biolinerx-reports-third-quarter-2014-120000713.html
$29.6 million in cash
http://finance.yahoo.com/news/biolinerx-reports-third-quarter-2014-120000713.html
$29.6 million in cash
raggirays
Utente Registrato
- Registrato
- 7/4/10
- Messaggi
- 828
- Punti reazioni
- 36
Form 6-K BioLineRx Ltd. For: Dec 08
BioLineRx Reports Positive Data from Ongoing Phase 2a
Study for AML Treatment at ASH conference
- Data show six-fold increase in mobilization of AML cells from bone marrow;
Treatment with BL-8040 as single agent led to 70% decrease in AML cells in
bone marrow and 3.5-fold increase in AML cell apoptosis -
Jerusalem, Israel, December 8, 2014 – BioLineRx Ltd. (NASDAQ: BLRX; TASE: BLRX), a clinical-stage biopharmaceutical company dedicated to identifying, in-licensing and developing promising therapeutic candidates, announced today that data from the on-going Phase 2a clinical trial of BL-8040 for the treatment of patients with relapsed or refractory acute myeloid leukemia (AML) were reported at the American Society of Hematology (ASH) meeting, held on December 6-9, 2014, in San Francisco.
Results reported to date in the dose-escalation stage of the Phase 2a study show that, even at the highest dose reached to date (1.25 mg/kg), there were no dose-limiting toxicity events or serious adverse events, nor early discontinuations attributable to BL-8040. Furthermore, BL-8040 triggered substantial mobilization of AML cancer cells from the bone marrow to the peripheral blood, with a median 6-fold increase of AML cells in the blood. This mobilization is crucial for exposing a higher ratio of AML cells to accompanying chemotherapy such as Ara-C. Additional results show that after only two days of BL-8040 monotherapy, there was a median decrease of approximately 70% in the amount of AML cells in the bone marrow, while the levels of normal progenitor cells remained stable. Furthermore, BL-8040 as a monotherapy showed a 3.5-fold increase in cell death (apoptosis) of AML cells, both in the bone marrow and in peripheral blood samples.
Dr. Kinneret Savitsky, Chief Executive Officer of BioLineRx, stated, “We are very encouraged by the data that we see at this stage of the Phase 2a study for BL-8040, which show substantial mobilization and robust apoptosis, and we hope that we will continue to see a dose response as we test higher doses. The dose escalation stage, which is currently ongoing, is expected to be completed early next year. In addition, we recently added the Mayo Clinic as our fourth world-class site in the U.S., and we plan to open up several additional sites in the U.S. in the next few months. We look forward to reporting results of the escalation stage, and initiating the expansion phase of the trial, in which the optimal dose of BL-8040 will be further assessed for safety as well as efficacy. The full study is expected to be completed in the second half of 2015.”
BioLineRx Reports Positive Data from Ongoing Phase 2a
Study for AML Treatment at ASH conference
- Data show six-fold increase in mobilization of AML cells from bone marrow;
Treatment with BL-8040 as single agent led to 70% decrease in AML cells in
bone marrow and 3.5-fold increase in AML cell apoptosis -
Jerusalem, Israel, December 8, 2014 – BioLineRx Ltd. (NASDAQ: BLRX; TASE: BLRX), a clinical-stage biopharmaceutical company dedicated to identifying, in-licensing and developing promising therapeutic candidates, announced today that data from the on-going Phase 2a clinical trial of BL-8040 for the treatment of patients with relapsed or refractory acute myeloid leukemia (AML) were reported at the American Society of Hematology (ASH) meeting, held on December 6-9, 2014, in San Francisco.
Results reported to date in the dose-escalation stage of the Phase 2a study show that, even at the highest dose reached to date (1.25 mg/kg), there were no dose-limiting toxicity events or serious adverse events, nor early discontinuations attributable to BL-8040. Furthermore, BL-8040 triggered substantial mobilization of AML cancer cells from the bone marrow to the peripheral blood, with a median 6-fold increase of AML cells in the blood. This mobilization is crucial for exposing a higher ratio of AML cells to accompanying chemotherapy such as Ara-C. Additional results show that after only two days of BL-8040 monotherapy, there was a median decrease of approximately 70% in the amount of AML cells in the bone marrow, while the levels of normal progenitor cells remained stable. Furthermore, BL-8040 as a monotherapy showed a 3.5-fold increase in cell death (apoptosis) of AML cells, both in the bone marrow and in peripheral blood samples.
Dr. Kinneret Savitsky, Chief Executive Officer of BioLineRx, stated, “We are very encouraged by the data that we see at this stage of the Phase 2a study for BL-8040, which show substantial mobilization and robust apoptosis, and we hope that we will continue to see a dose response as we test higher doses. The dose escalation stage, which is currently ongoing, is expected to be completed early next year. In addition, we recently added the Mayo Clinic as our fourth world-class site in the U.S., and we plan to open up several additional sites in the U.S. in the next few months. We look forward to reporting results of the escalation stage, and initiating the expansion phase of the trial, in which the optimal dose of BL-8040 will be further assessed for safety as well as efficacy. The full study is expected to be completed in the second half of 2015.”