Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
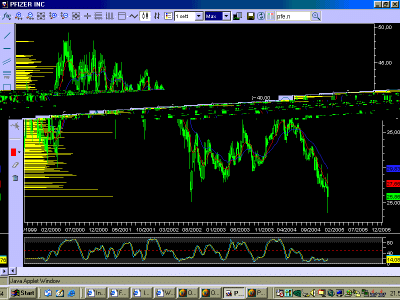
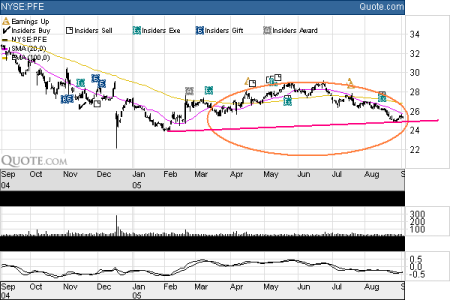
PFE - Pfizer Inc. (NYSE:PFE)
- Creatore Discussione poncio
- Data di inizio
Più opzioni
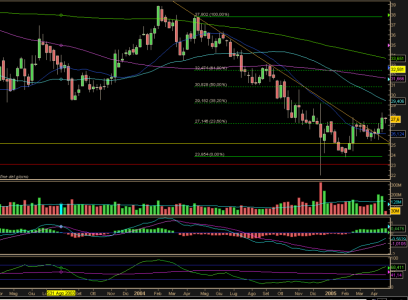
Chi ha risposto?poncio ha scritto:Grafico weekly.
La scoppola odierna l'ha riportato sui minimi di medio, a 25,85 c'è un livello di market profile.
La stavo guardando adesso,a questi prezzi è sicuramente sottovalutata,,,,
mi piace anche a me.....
vi fate un giro sulla spaiggia nei momenti di affollamento da voi e potete tranquillamente fare a meno dei prodotti di questa aziendaponcio ha scritto:Poi per noi anziani ha un certo valore intrinseco...

skepsi
Nuovo Utente
- Registrato
- 22/1/02
- Messaggi
- 9.691
- Punti reazioni
- 727
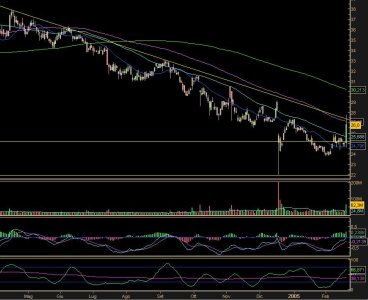
Personalmente la faccenda non mi è piaciuta affatto.
Non solo perché ho visto svalutato il mio investimento, ma perché trattasi del IV ribasso consecutivo e con volumi stratosferici.
Il titolo è partito da un minimo di 21,99$ quando ieri aveva concluso la seduta a 29$.
Un convegno organizzato dall’Istituto di ricerca sul cancro che sortisce questi effetti su un azienda è una specie di catastrofe in un microcosmo aziendale.
Personalmente attendo di vedere dopo questa specie di recupero odierno sia quanto si recupererà e in quanto tempo.
Non credo che i tempi saranno rapidi.
Quando si opera in questo modo i mesi che possono trascorrere per conseguire anche parziali recuperi, potrebbero avere le sembianze dell’eternità.
Volatilità storica un'altra volta verso i massimi. Migliaia di contratti sulle opzioni che indicano chiaramente che la festa andrà avanti.
Non solo perché ho visto svalutato il mio investimento, ma perché trattasi del IV ribasso consecutivo e con volumi stratosferici.
Il titolo è partito da un minimo di 21,99$ quando ieri aveva concluso la seduta a 29$.
Un convegno organizzato dall’Istituto di ricerca sul cancro che sortisce questi effetti su un azienda è una specie di catastrofe in un microcosmo aziendale.
Personalmente attendo di vedere dopo questa specie di recupero odierno sia quanto si recupererà e in quanto tempo.
Non credo che i tempi saranno rapidi.
Quando si opera in questo modo i mesi che possono trascorrere per conseguire anche parziali recuperi, potrebbero avere le sembianze dell’eternità.
Volatilità storica un'altra volta verso i massimi. Migliaia di contratti sulle opzioni che indicano chiaramente che la festa andrà avanti.
scusate ho bisogno di una mano,
avevo inserito un ordine sul titolo a 23,6 alle ore 15,53 (in quel momento prezzava a 23,5) il titolo e' rimasto per 3-4 minuti sotto i 23,4 ma non ho avuto l'eseguito, mi risulta e ne sono certo che non sia l'unico caso,
ho chiamato in banca hanno detto che verificheranno lunedi...secondo voi che posso fare.....ciao e grazie
avevo inserito un ordine sul titolo a 23,6 alle ore 15,53 (in quel momento prezzava a 23,5) il titolo e' rimasto per 3-4 minuti sotto i 23,4 ma non ho avuto l'eseguito, mi risulta e ne sono certo che non sia l'unico caso,
ho chiamato in banca hanno detto che verificheranno lunedi...secondo voi che posso fare.....ciao e grazie
skepsi
Nuovo Utente
- Registrato
- 22/1/02
- Messaggi
- 9.691
- Punti reazioni
- 727
Credo che ad inizio seduta c'è stata una parziale sospensione, considerata sia l'entità del ribasso che i volumi.
Possibile che il tuo ordine non sia stato eseguito.
Gli ordini non erano eseguibili da diversi TOL ad inizio seduta.
Comunque cerca di insistere con la tua banca e stampa la proposta da cui si rileva l'orario di trasmissione e immissione dell'ordine.
Possibile che il tuo ordine non sia stato eseguito.
Gli ordini non erano eseguibili da diversi TOL ad inizio seduta.
Comunque cerca di insistere con la tua banca e stampa la proposta da cui si rileva l'orario di trasmissione e immissione dell'ordine.
simulpaolo
Gold is money
- Registrato
- 26/11/00
- Messaggi
- 22.571
- Punti reazioni
- 524
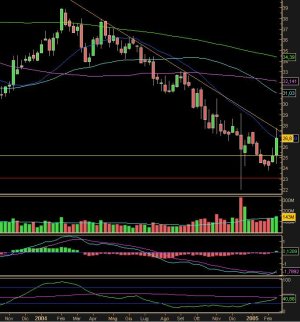
Il falling knife non fa certo al caso mio, questa potrebbe scendere anche sotto i 20$ con i successivi downgrade e processi legali.
Avete visto anche Astrazeneca che batosta oggi.
Non è tempo di big del pharma ora, meglio aspettare la prosima primavera .
Paolo
Avete visto anche Astrazeneca che batosta oggi.
Non è tempo di big del pharma ora, meglio aspettare la prosima primavera .
Paolo
Pfizer sarà buona da comprare per il cassetto fra un mese circa dopo la stabilizzazione dei prezzi e del danno dovuto al probabile ritiro del farmaco; è gia successo con Merck ed è stato un affare ma per cambiare campo è successo anche con Nokia che dopo un mese o poco più dalle batoste sui profit w. è poi risalita molto bene.
Merck e Pfizer a questi prezzi possono essere un affare anche per il rischio di una opa ostile .
Ciao a tutti e Buon Natale
Merck e Pfizer a questi prezzi possono essere un affare anche per il rischio di una opa ostile .
Ciao a tutti e Buon Natale
incominciano...
Investor Notice: Murray, Frank & Sailer LLP Has Filed A Shareholder Class Action [DSZMCMF]
NEW YORK, Dec. 17, 2004 (PRIMEZONE) -- Murray, Frank & Sailer LLP has filed a
class action lawsuit on behalf of shareholders who purchased or otherwise
acquired the securities of Pfizer, Inc. ("Pfizer" or the "Company") (NYSE FE)
FE)
between November 1, 2000 and December 16, 2004, inclusive (the "Class Period").
The complaint alleges that defendants misrepresented and omitted material facts
about the safety and marketability of Pfizer's Celebrex and Bextra products.
Plaintiff further alleges that Defendants were aware of strong indications that
Celebrex and Bextra, drugs known as "Cox-2 Inhibitors," posed serious and
undisclosed health risks to the drug's consumers, that the undisclosed health
risks would hinder their marketability, and that the potential financial
liability Pfizer would face due to these drugs' harms posed a serious financial
threat to the Company. Despite such knowledge, Defendants continued to conceal
these facts from consumers and the investing public.
A series of revelations caused the market to learn the truth about Bextra and
Celebrex. On November 4, 2004, the Calgary Herald reported that "Celebrex, a
popular pain drug touted as the safe alternative after Vioxx was pulled from
drugstore shelves, is suspected of causing at least 14 deaths and numerous heart
and brain side effects." Then, on November 10, 2004, the New York Times revealed
a study finding that "(t)he incidence of heart attacks and strokes among
patients given Pfizer's painkiller Bextra was more than double that of those
given placebos." This news shocked the market, causing Pfizer's share price to
drop 8% over the next eight days.
Before the market opened today, Pfizer again shocked the market, revealing that
"(i)n the Adenoma Prevention with Celecoxib (APC) trial, patients taking 400mg
and 800mg of Celebrex daily had an approximately 2.5 fold increase in their risk
of experiencing a major fatal or non-fatal cardiovascular event compared to
those patients taking placebo, according to the National Cancer Institute (NCI).
Based on these statistically significant findings, the sponsor of the trial, the
NCI, has suspended the dosing of Celebrex in the study." Pfizer's share price
has dropped precipitously in response to this news.
Murray, Frank & Sailer LLP and its predecessor firms have devoted its practice
to shareholder class actions and complex commercial litigation for more than
thirty years and have recovered hundreds of millions of dollars for shareholders
in class actions throughout the United States.
If you purchased (on the open market or in an employee retirement account) or
otherwise acquired Pfizer securities on any world exchange between November 1,
2000 and December 16, 2004, and sustained damages, you may, no later than
February 14, 2005, move the Court to serve as lead plaintiff. Shareholders
outside the United States may also join the action, regardless of which exchange
was used to purchase the securities. To serve as lead plaintiff, however, you
must meet certain legal requirements. You can join this class action as lead
plaintiff online at http://www.murrayfrank.com/CM/NewCases/NewCases.asp. If you
would like to discuss this action, this announcement, or your rights and
interests, please contact plaintiff's counsel Eric J. Belfi or Aaron D. Patton
of Murray, Frank & Sailer LLP.
More information on this and other class actions can be found on the Class
Action Newsline at www.primezone.com/ca
Investor Notice: Murray, Frank & Sailer LLP Has Filed A Shareholder Class Action [DSZMCMF]
NEW YORK, Dec. 17, 2004 (PRIMEZONE) -- Murray, Frank & Sailer LLP has filed a
class action lawsuit on behalf of shareholders who purchased or otherwise
acquired the securities of Pfizer, Inc. ("Pfizer" or the "Company") (NYSE
between November 1, 2000 and December 16, 2004, inclusive (the "Class Period").
The complaint alleges that defendants misrepresented and omitted material facts
about the safety and marketability of Pfizer's Celebrex and Bextra products.
Plaintiff further alleges that Defendants were aware of strong indications that
Celebrex and Bextra, drugs known as "Cox-2 Inhibitors," posed serious and
undisclosed health risks to the drug's consumers, that the undisclosed health
risks would hinder their marketability, and that the potential financial
liability Pfizer would face due to these drugs' harms posed a serious financial
threat to the Company. Despite such knowledge, Defendants continued to conceal
these facts from consumers and the investing public.
A series of revelations caused the market to learn the truth about Bextra and
Celebrex. On November 4, 2004, the Calgary Herald reported that "Celebrex, a
popular pain drug touted as the safe alternative after Vioxx was pulled from
drugstore shelves, is suspected of causing at least 14 deaths and numerous heart
and brain side effects." Then, on November 10, 2004, the New York Times revealed
a study finding that "(t)he incidence of heart attacks and strokes among
patients given Pfizer's painkiller Bextra was more than double that of those
given placebos." This news shocked the market, causing Pfizer's share price to
drop 8% over the next eight days.
Before the market opened today, Pfizer again shocked the market, revealing that
"(i)n the Adenoma Prevention with Celecoxib (APC) trial, patients taking 400mg
and 800mg of Celebrex daily had an approximately 2.5 fold increase in their risk
of experiencing a major fatal or non-fatal cardiovascular event compared to
those patients taking placebo, according to the National Cancer Institute (NCI).
Based on these statistically significant findings, the sponsor of the trial, the
NCI, has suspended the dosing of Celebrex in the study." Pfizer's share price
has dropped precipitously in response to this news.
Murray, Frank & Sailer LLP and its predecessor firms have devoted its practice
to shareholder class actions and complex commercial litigation for more than
thirty years and have recovered hundreds of millions of dollars for shareholders
in class actions throughout the United States.
If you purchased (on the open market or in an employee retirement account) or
otherwise acquired Pfizer securities on any world exchange between November 1,
2000 and December 16, 2004, and sustained damages, you may, no later than
February 14, 2005, move the Court to serve as lead plaintiff. Shareholders
outside the United States may also join the action, regardless of which exchange
was used to purchase the securities. To serve as lead plaintiff, however, you
must meet certain legal requirements. You can join this class action as lead
plaintiff online at http://www.murrayfrank.com/CM/NewCases/NewCases.asp. If you
would like to discuss this action, this announcement, or your rights and
interests, please contact plaintiff's counsel Eric J. Belfi or Aaron D. Patton
of Murray, Frank & Sailer LLP.
More information on this and other class actions can be found on the Class
Action Newsline at www.primezone.com/ca
C' è anche questa di oggi.
CR: Eyetech and Pfizer Announce FDA Approval of Macugen(R) for Treatment of [DSZKBYD]
08:18pm EST 17-Dec-04 PR Newswire - First Call Wire (Eyetech Pharmaceuticals,
Eyetech and Pfizer Announce FDA Approval of Macugen(R) for Treatment of
Neovascular (Wet) Age-Related Macular Degeneration
- First treatment that helps preserve vision by targeting an underlying cause
of disease -
NEW YORK, Dec. 17 /PRNewswire-FirstCall/ -- Eyetech Pharmaceuticals, Inc.
(Nasdaq: EYET) and Pfizer Inc (NYSE: PFE) announced today that the U.S. Food
and Drug Administration (FDA) approved Macugen(R) (pegaptanib sodium
injection) for the treatment of neovascular (wet) age-related macular
degeneration (AMD), an eye disease associated with aging that destroys central
vision. AMD is the leading cause of irreversible severe vision loss in
patients older than 50 years of age in developed countries. Macugen helps
preserve vision and helps limit progression to legal blindness. Approval of
Macugen follows a priority review under the FDA's rolling submission-Pilot 1
program based on data from the companies' Phase 2/3 pivotal clinical trials.
Macugen is the first in a new class of ophthalmic drugs to specifically
target vascular endothelial growth factor (VEGF), a protein which acts as a
signal in triggering the abnormal blood vessel growth and leakage that is the
hallmark of neovascular AMD.
"Macugen is the first anti-angiogenic treatment approved in ophthalmology
and represents the beginning of a new era. The anti-angiogenic approach
specifically addresses, for the first time, an underlying cause of blindness
in age-related macular degeneration. Anti-angiogenesis has evolved from
theory to therapy," said Judah Folkman, M.D., Julia Andrus Dyckman Professor
of Pediatric Surgery at Children's Hospital in Boston and Harvard Medical
School.
CR: Eyetech and Pfizer Announce FDA Approval of Macugen(R) for Treatment of [DSZKBYD]
08:18pm EST 17-Dec-04 PR Newswire - First Call Wire (Eyetech Pharmaceuticals,
Eyetech and Pfizer Announce FDA Approval of Macugen(R) for Treatment of
Neovascular (Wet) Age-Related Macular Degeneration
- First treatment that helps preserve vision by targeting an underlying cause
of disease -
NEW YORK, Dec. 17 /PRNewswire-FirstCall/ -- Eyetech Pharmaceuticals, Inc.
(Nasdaq: EYET) and Pfizer Inc (NYSE: PFE) announced today that the U.S. Food
and Drug Administration (FDA) approved Macugen(R) (pegaptanib sodium
injection) for the treatment of neovascular (wet) age-related macular
degeneration (AMD), an eye disease associated with aging that destroys central
vision. AMD is the leading cause of irreversible severe vision loss in
patients older than 50 years of age in developed countries. Macugen helps
preserve vision and helps limit progression to legal blindness. Approval of
Macugen follows a priority review under the FDA's rolling submission-Pilot 1
program based on data from the companies' Phase 2/3 pivotal clinical trials.
Macugen is the first in a new class of ophthalmic drugs to specifically
target vascular endothelial growth factor (VEGF), a protein which acts as a
signal in triggering the abnormal blood vessel growth and leakage that is the
hallmark of neovascular AMD.
"Macugen is the first anti-angiogenic treatment approved in ophthalmology
and represents the beginning of a new era. The anti-angiogenic approach
specifically addresses, for the first time, an underlying cause of blindness
in age-related macular degeneration. Anti-angiogenesis has evolved from
theory to therapy," said Judah Folkman, M.D., Julia Andrus Dyckman Professor
of Pediatric Surgery at Children's Hospital in Boston and Harvard Medical
School.
Paolino967
L'AQUILA
- Registrato
- 7/1/01
- Messaggi
- 5.978
- Punti reazioni
- 176
Arba ha scritto:Pfizer sarà buona da comprare per il cassetto fra un mese circa dopo la stabilizzazione dei prezzi e del danno dovuto al probabile ritiro del farmaco; è gia successo con Merck ed è stato un affare ma per cambiare campo è successo anche con Nokia che dopo un mese o poco più dalle batoste sui profit w. è poi risalita molto bene.
Merck e Pfizer a questi prezzi possono essere un affare anche per il rischio di una opa ostile .
Ciao a tutti e Buon Natale
opa ostile da parte di chi??? hai visto quanto capitalizza PF???
Ngc-53
sunt tecum quae fugi
- Registrato
- 28/5/03
- Messaggi
- 2.719
- Punti reazioni
- 125
Ngc-53
sunt tecum quae fugi
- Registrato
- 28/5/03
- Messaggi
- 2.719
- Punti reazioni
- 125
Ngc-53
sunt tecum quae fugi
- Registrato
- 28/5/03
- Messaggi
- 2.719
- Punti reazioni
- 125
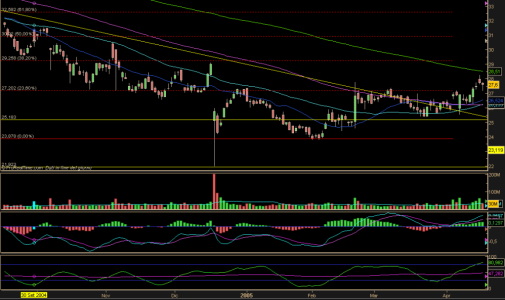
usciti i dati, 1 cdnts meglio delle previsioni e un outlook positivo, resta da vedere come reagirà il mercato.
http://www.thestreet.com/_yahoo/stocks/biotech/10218266.html?cm_ven=YAHOO&cm_cat=FREE&cm_ite=NA
http://www.thestreet.com/_yahoo/stocks/biotech/10218266.html?cm_ven=YAHOO&cm_cat=FREE&cm_ite=NA