Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
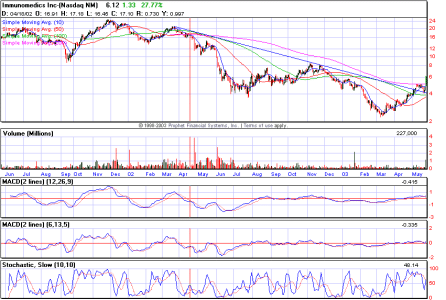
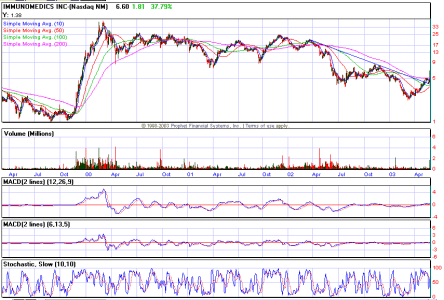
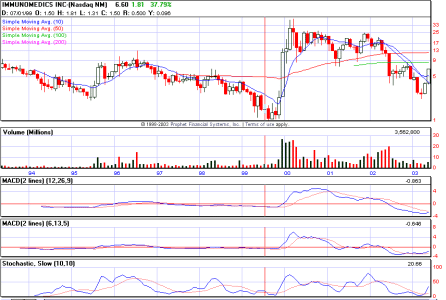
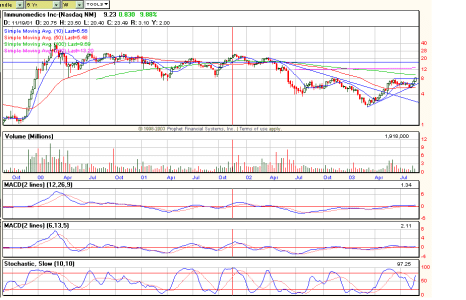
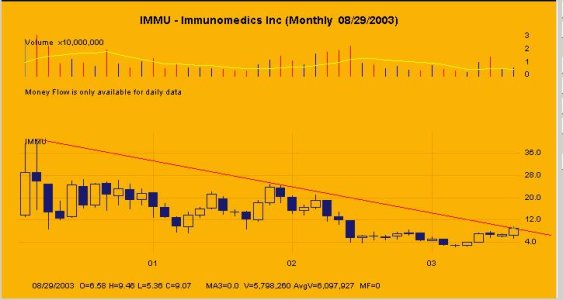
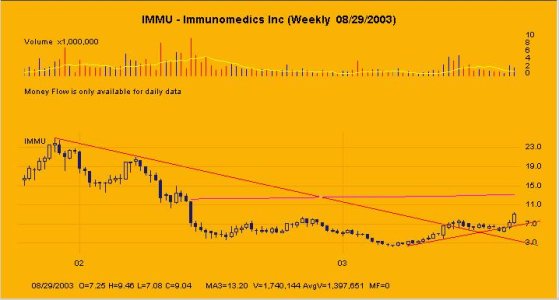
IMMU (Da Paura)
- Creatore Discussione BigNick
- Data di inizio
Più opzioni
Chi ha risposto?MaxVaro
Nuovo Utente
- Registrato
- 26/2/01
- Messaggi
- 3.970
- Punti reazioni
- 302
Ciao Big! 
Non la seguo ma in effetti non è male il grafico.... anzi !
Visto ke roba SIRI ?! avevo paura che sciacquasse giù prima di ripartire invece l'effetto supp. a 1$ ha funzionato molto bene...
Ci sent. nelle prossime sere, tra poco vado a spasso.... in Italy non male ma lascio sempre sul tavolo le operaz. migliori !!!
Buona Serata, Max
Non la seguo ma in effetti non è male il grafico.... anzi !
Visto ke roba SIRI ?! avevo paura che sciacquasse giù prima di ripartire invece l'effetto supp. a 1$ ha funzionato molto bene...
Ci sent. nelle prossime sere, tra poco vado a spasso.... in Italy non male ma lascio sempre sul tavolo le operaz. migliori !!!
Buona Serata, Max
BigNick
Siamo solo noi
- Registrato
- 10/7/01
- Messaggi
- 12.147
- Punti reazioni
- 1.266
r
ciao Max, guardati anche ONXX e BIOM
aspetto tue notizie
BigNIck
Scritto da MaxVaro
Ciao Big!
Non la seguo ma in effetti non è male il grafico.... anzi !
Visto ke roba SIRI ?! avevo paura che sciacquasse giù prima di ripartire invece l'effetto supp. a 1$ ha funzionato molto bene...
Ci sent. nelle prossime sere, tra poco vado a spasso.... in Italy non male ma lascio sempre sul tavolo le operaz. migliori !!!
Buona Serata, Max
ciao Max, guardati anche ONXX e BIOM
aspetto tue notizie
BigNIck
BigNick
Siamo solo noi
- Registrato
- 10/7/01
- Messaggi
- 12.147
- Punti reazioni
- 1.266
Re: Non mola, 6,90 shorters
niente
BigNick
Scritto da BigNick
vediamo se chi l'ha shortata si ricopre prima della fine
BigNick
niente
BigNick
BigNick
Siamo solo noi
- Registrato
- 10/7/01
- Messaggi
- 12.147
- Punti reazioni
- 1.266
Immunomedics Completes NJEDA Bond Financing for Large-Scale Manufacturing Facility
Source: Immunomedics, Inc.
Immunomedics Completes NJEDA Bond Financing for Large-Scale Manufacturing Facility
Wednesday May 28, 12:57 pm ET
MORRIS PLAINS, N.J., May 28 /PRNewswire-FirstCall/ -- Immunomedics, Inc. (Nasdaq: IMMU - News) today announced that it has successfully completed a $6.4 million Bond Financing with the New Jersey Economic Development Authority ("NJEDA"). The proceeds, which are to be repaid over a five-year period, will be used to refinance the expansion of the Company's manufacturing facility in Morris Plains, N.J. Mr. Gerard G. Gorman, the Company's Vice President, Finance, and Chief Financial Officer, commented, "We are very excited about working with the NJEDA and appreciate their help in financing the expansion of our facility. With this financing, our cash position remains strong at approximately $33.0 million."
Immunomedics' President and Chief Executive Officer, Ms. Cynthia L. Sullivan, remarked: "The expansion financed by these bonds will enable Immunomedics to increase our bioreactor manufacturing capacity in New Jersey to support all of our clinical trial requirements for today, as well as potential commercial quantities of future antibody-based cancer therapeutic products."
"Low-cost EDA bond financing is an effective tool for qualifying New Jersey manufacturers to borrow for projects like building acquisitions or equipment purchases," said EDA Executive Director, Ms. Caren S. Franzini. "Providing high-tech businesses like Immunomedics with the capital to support their growth is a key element in Governor McGreevey's strategies for building a solid economic future and keeping New Jersey at the forefront of scientific discovery and development."
Immunomedics is a biopharmaceutical company focused on the development, manufacture and commercialization of diagnostic imaging and therapeutic products for the detection and treatment of cancer and infectious diseases. Integral to these products are highly specific monoclonal antibodies and antibody fragments designed to deliver radioisotopes and chemotherapeutic agents to tumors and sites of infection. Immunomedics has six therapeutic product candidates in clinical trials and has two marketed diagnostic imaging products. The most advanced therapeutic product candidates are LymphoCide® (epratuzumab), which is in Phase II clinical trials for the treatment of non-Hodgkin's lymphoma, and CEA-Cide® (labetuzumab), which is in Phase I/II clinical trials for the treatment of certain solid tumors.
This release, in addition to historical information, contains forward-looking statements made pursuant to the Private Securities Litigation Reform Act of 1995. Such statements, including statements regarding clinical trials, involve significant risks and uncertainties and actual results could differ materially from those expressed or implied herein. Factors that could cause such differences include, but are not limited to, risks associated with new product development (including clinical trials outcome and regulatory requirements/actions), competitive risks to marketed products and availability of financing and other sources of capital, as well as the risks discussed in the Company's Annual Report on Form 10-K for the year June 30, 2002.
Source: Immunomedics, Inc.
Immunomedics Completes NJEDA Bond Financing for Large-Scale Manufacturing Facility
Wednesday May 28, 12:57 pm ET
MORRIS PLAINS, N.J., May 28 /PRNewswire-FirstCall/ -- Immunomedics, Inc. (Nasdaq: IMMU - News) today announced that it has successfully completed a $6.4 million Bond Financing with the New Jersey Economic Development Authority ("NJEDA"). The proceeds, which are to be repaid over a five-year period, will be used to refinance the expansion of the Company's manufacturing facility in Morris Plains, N.J. Mr. Gerard G. Gorman, the Company's Vice President, Finance, and Chief Financial Officer, commented, "We are very excited about working with the NJEDA and appreciate their help in financing the expansion of our facility. With this financing, our cash position remains strong at approximately $33.0 million."
Immunomedics' President and Chief Executive Officer, Ms. Cynthia L. Sullivan, remarked: "The expansion financed by these bonds will enable Immunomedics to increase our bioreactor manufacturing capacity in New Jersey to support all of our clinical trial requirements for today, as well as potential commercial quantities of future antibody-based cancer therapeutic products."
"Low-cost EDA bond financing is an effective tool for qualifying New Jersey manufacturers to borrow for projects like building acquisitions or equipment purchases," said EDA Executive Director, Ms. Caren S. Franzini. "Providing high-tech businesses like Immunomedics with the capital to support their growth is a key element in Governor McGreevey's strategies for building a solid economic future and keeping New Jersey at the forefront of scientific discovery and development."
Immunomedics is a biopharmaceutical company focused on the development, manufacture and commercialization of diagnostic imaging and therapeutic products for the detection and treatment of cancer and infectious diseases. Integral to these products are highly specific monoclonal antibodies and antibody fragments designed to deliver radioisotopes and chemotherapeutic agents to tumors and sites of infection. Immunomedics has six therapeutic product candidates in clinical trials and has two marketed diagnostic imaging products. The most advanced therapeutic product candidates are LymphoCide® (epratuzumab), which is in Phase II clinical trials for the treatment of non-Hodgkin's lymphoma, and CEA-Cide® (labetuzumab), which is in Phase I/II clinical trials for the treatment of certain solid tumors.
This release, in addition to historical information, contains forward-looking statements made pursuant to the Private Securities Litigation Reform Act of 1995. Such statements, including statements regarding clinical trials, involve significant risks and uncertainties and actual results could differ materially from those expressed or implied herein. Factors that could cause such differences include, but are not limited to, risks associated with new product development (including clinical trials outcome and regulatory requirements/actions), competitive risks to marketed products and availability of financing and other sources of capital, as well as the risks discussed in the Company's Annual Report on Form 10-K for the year June 30, 2002.
okkrim
semprepinze
- Registrato
- 10/6/01
- Messaggi
- 2.381
- Punti reazioni
- 153
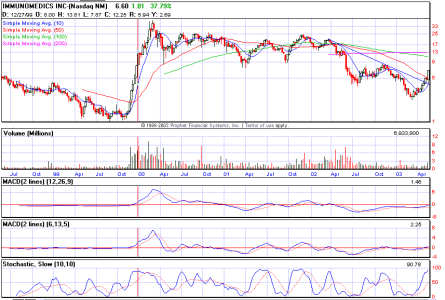
Re: se torna a 8,7..
assolutamente fantastica BIG
Scritto da BigNick
potrei anche provare a prenderne un po'
assolutamente fantastica BIG
Allegati
okkrim
semprepinze
- Registrato
- 10/6/01
- Messaggi
- 2.381
- Punti reazioni
- 153
Re: piace anche a me...
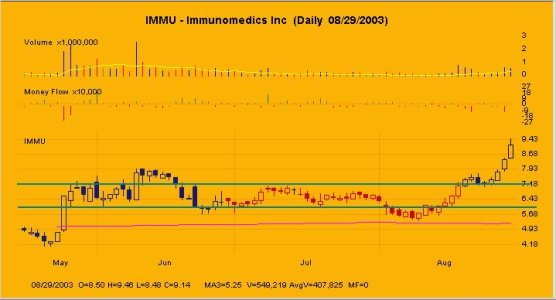
beh... a mio giudizio facile che storni...
sul D sta formando una shooting ed ha climactic volume... indice ke benzina finita...
potrei sbagliare ma se le avessi sarei gia' fuori...
non avendole attendo paziente...
Questa fara' faville, a mio giudizio, ma non ora
Sempre pinze....
Ciao
Scritto da BigNick
però non molla !
che dici viene giù ?
per me questa sera non scende sotto gli 8,9$
BigNick
beh... a mio giudizio facile che storni...
sul D sta formando una shooting ed ha climactic volume... indice ke benzina finita...
potrei sbagliare ma se le avessi sarei gia' fuori...
non avendole attendo paziente...
Questa fara' faville, a mio giudizio, ma non ora
Sempre pinze....
Ciao
BigNick
Siamo solo noi
- Registrato
- 10/7/01
- Messaggi
- 12.147
- Punti reazioni
- 1.266
Re: Re: piace anche a me...
secondo te quale potrebbe essere una buona entrata ?
BigNick
Scritto da okkrim
beh... a mio giudizio facile che storni...
sul D sta formando una shooting ed ha climactic volume... indice ke benzina finita...
potrei sbagliare ma se le avessi sarei gia' fuori...
non avendole attendo paziente...
Questa fara' faville, a mio giudizio, ma non ora
Sempre pinze....
Ciao
secondo te quale potrebbe essere una buona entrata ?
BigNick
okkrim
semprepinze
- Registrato
- 10/6/01
- Messaggi
- 2.381
- Punti reazioni
- 153
Re: Re: Re: piace anche a me...
non ne ho la piu' pallida idea Big
direi di attendere una candela di inversione
quella di oggi conferma quella di venerdi...
a mio giudizio deve scaricare un poko
fra 7,95 e 8,40 c'e' un'area di forte supporto se non buca la', facile ke la ripartenza sia a breve...
ma a quanto non lo so...
ciao
Scritto da BigNick
secondo te quale potrebbe essere una buona entrata ?
BigNick
non ne ho la piu' pallida idea Big
direi di attendere una candela di inversione
quella di oggi conferma quella di venerdi...
a mio giudizio deve scaricare un poko
fra 7,95 e 8,40 c'e' un'area di forte supporto se non buca la', facile ke la ripartenza sia a breve...
ma a quanto non lo so...
ciao