Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Acasti Pharma Inc(NASDAQ:ACST) pronta al botto!!!!
- Creatore Discussione dariope
- Data di inizio
-
 Trading Day 19 aprile Torino - Corso Gratuito sull'investimento
Trading Day 19 aprile Torino - Corso Gratuito sull'investimento

Migliora la tua strategia di trading con le preziose intuizioni dei nostri esperti su oro, materie prime, analisi tecnica, criptovalute e molto altro ancora. Iscriviti subito per partecipare gratuitamente allo Swissquote Trading Day.
Per continuare a leggere visita questo LINK
Più opzioni
Chi ha risposto?renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
forse vale un cippino.....anche i morti a volte resuscitano 
LAVAL, QUEBEC--(Marketwired - Sep 14, 2016) - Acasti Pharma (ACST)(TSX VENTURE:APO) today announced that its bridging study for novel drug candidate CaPre® (omega-3 phospholipid) has successfully met its objectives, supporting Acasti's strategy to pursue the U.S. Food and Drug Administration's (FDA) 505(b)(2) regulatory pathway for approval. Acasti is developing CaPre for the treatment of patients with severe hypertriglyceridemia, a metabolic condition that contributes to increased risk of cardiovascular disease and pancreatitis. The 505(b)(2) regulatory pathway allows Acasti to streamline the overall development program required to support a New Drug Application (NDA) by relying on the safety data of an approved drug.
"We are confident that the results of this study support the 505(b)(2) regulatory pathway chosen by Acasti to gain marketing approval of CaPre," said Jan D'Alvise, president and CEO of Acasti Pharma. "With this momentum, we look forward to working with the FDA to confirm the pathway and optimize the design of our Phase 3 program, which will seek to demonstrate the safety and efficacy of CaPre in patients with severe hypertriglyceridemia."
Acasti's open-label, randomized, four-way, cross-over, bioavailability study compared CaPre given as a single dose of 4 grams in fasting and fed states with the approved hypertriglyceridemia drug LOVAZA (omega-3-acid ethyl esters) in 56 healthy volunteers. The study met its primary objective and demonstrated that the levels of omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) following administration of CaPre did not exceed the levels following administration of LOVAZA in subjects who were fed a high-fat meal. These results support the basis for claiming a comparable safety profile of the two products.
Furthermore, among subjects in the fasting state, CaPre demonstrated better bioavailability than LOVAZA, as measured by blood levels of EPA and DHA. As previously reported, the bioavailability of CaPre is not significantly reduced when taken with a low-fat meal versus a high-fat meal. This could represent a significant clinical advantage for CaPre since the administration with a low-fat meal represents a more attractive regimen for patients with hypertriglyceridemia who follow a restricted diet.
CaPre is a novel composition of omega-3 phospholipids sourced from krill. Its omega-3s, principally EPA and DHA, are naturally either "free" or bound to phospholipids that help them to be better absorbed into the body. This allows for enhanced bioavailability and EPA and DHA blood levels compared to the "esterified" fish-oil omega-3 options such as LOVAZA. CaPre is designed to modulate the major lipids associated with cardio-metabolic disease: in two previously reported Phase 2 clinical trials, CaPre reduced triglyceride levels, lowered non-high density lipoprotein (non-HDL-C, a useful marker of cardiovascular disease), and increased levels of high density lipoprotein (HDL-C, or "good cholesterol") while having a neutral to positive effect on lowering low density lipoprotein (LDL-C, or "bad cholesterol").
"The CaPre bioavailability study has reinforced the compound's unique attributes in comparison with the leading pharmaceutical agent for hypertriglyceridemia," said Roderick Carter, M.D., chairman of Acasti Pharma. "CaPre demonstrates clinically meaningful effects on many key markers of cardio-metabolic health. With high rates of obesity and diabetes fueling the number of patients with elevated triglycerides and cholesterols, CaPre could fill the need for a best-in-class omega-3 medication that addresses the full lipid profiles of these patients."
About CaPre
Sourced from krill oil, CaPre seeks to provide a full scope of health benefits to patients with hypertriglyceridemia, filling a medical need that no other omega-3 treatment option has been able to address. CaPre successfully completed Phase 2 clinical trials for the treatment of hypertriglyceridemia, a very common metabolic condition in which blood levels of triglycerides, a type of lipid, are elevated, posing a risk to cardiovascular health. Severe hypertriglyceridemia, affecting more than 4 million adults in the U.S. is associated with an increased risk of coronary artery disease and pancreatitis and is often caused or exacerbated by uncontrolled diabetes mellitus, obesity and sedentary habits. CaPre is intended to be taken orally once per day in capsule form.
LAVAL, QUEBEC--(Marketwired - Sep 14, 2016) - Acasti Pharma (ACST)(TSX VENTURE:APO) today announced that its bridging study for novel drug candidate CaPre® (omega-3 phospholipid) has successfully met its objectives, supporting Acasti's strategy to pursue the U.S. Food and Drug Administration's (FDA) 505(b)(2) regulatory pathway for approval. Acasti is developing CaPre for the treatment of patients with severe hypertriglyceridemia, a metabolic condition that contributes to increased risk of cardiovascular disease and pancreatitis. The 505(b)(2) regulatory pathway allows Acasti to streamline the overall development program required to support a New Drug Application (NDA) by relying on the safety data of an approved drug.
"We are confident that the results of this study support the 505(b)(2) regulatory pathway chosen by Acasti to gain marketing approval of CaPre," said Jan D'Alvise, president and CEO of Acasti Pharma. "With this momentum, we look forward to working with the FDA to confirm the pathway and optimize the design of our Phase 3 program, which will seek to demonstrate the safety and efficacy of CaPre in patients with severe hypertriglyceridemia."
Acasti's open-label, randomized, four-way, cross-over, bioavailability study compared CaPre given as a single dose of 4 grams in fasting and fed states with the approved hypertriglyceridemia drug LOVAZA (omega-3-acid ethyl esters) in 56 healthy volunteers. The study met its primary objective and demonstrated that the levels of omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) following administration of CaPre did not exceed the levels following administration of LOVAZA in subjects who were fed a high-fat meal. These results support the basis for claiming a comparable safety profile of the two products.
Furthermore, among subjects in the fasting state, CaPre demonstrated better bioavailability than LOVAZA, as measured by blood levels of EPA and DHA. As previously reported, the bioavailability of CaPre is not significantly reduced when taken with a low-fat meal versus a high-fat meal. This could represent a significant clinical advantage for CaPre since the administration with a low-fat meal represents a more attractive regimen for patients with hypertriglyceridemia who follow a restricted diet.
CaPre is a novel composition of omega-3 phospholipids sourced from krill. Its omega-3s, principally EPA and DHA, are naturally either "free" or bound to phospholipids that help them to be better absorbed into the body. This allows for enhanced bioavailability and EPA and DHA blood levels compared to the "esterified" fish-oil omega-3 options such as LOVAZA. CaPre is designed to modulate the major lipids associated with cardio-metabolic disease: in two previously reported Phase 2 clinical trials, CaPre reduced triglyceride levels, lowered non-high density lipoprotein (non-HDL-C, a useful marker of cardiovascular disease), and increased levels of high density lipoprotein (HDL-C, or "good cholesterol") while having a neutral to positive effect on lowering low density lipoprotein (LDL-C, or "bad cholesterol").
"The CaPre bioavailability study has reinforced the compound's unique attributes in comparison with the leading pharmaceutical agent for hypertriglyceridemia," said Roderick Carter, M.D., chairman of Acasti Pharma. "CaPre demonstrates clinically meaningful effects on many key markers of cardio-metabolic health. With high rates of obesity and diabetes fueling the number of patients with elevated triglycerides and cholesterols, CaPre could fill the need for a best-in-class omega-3 medication that addresses the full lipid profiles of these patients."
About CaPre
Sourced from krill oil, CaPre seeks to provide a full scope of health benefits to patients with hypertriglyceridemia, filling a medical need that no other omega-3 treatment option has been able to address. CaPre successfully completed Phase 2 clinical trials for the treatment of hypertriglyceridemia, a very common metabolic condition in which blood levels of triglycerides, a type of lipid, are elevated, posing a risk to cardiovascular health. Severe hypertriglyceridemia, affecting more than 4 million adults in the U.S. is associated with an increased risk of coronary artery disease and pancreatitis and is often caused or exacerbated by uncontrolled diabetes mellitus, obesity and sedentary habits. CaPre is intended to be taken orally once per day in capsule form.
forse vale un cippino.....anche i morti a volte resuscitano
LAVAL, QUEBEC--(Marketwired - Sep 14, 2016) - Acasti Pharma (ACST)(TSX VENTURE:APO) today announced that its bridging study for novel drug candidate CaPre® (omega-3 phospholipid) has successfully met its objectives, supporting Acasti's strategy to pursue the U.S. Food and Drug Administration's (FDA) 505(b)(2) regulatory pathway for approval. Acasti is developing CaPre for the treatment of patients with severe hypertriglyceridemia, a metabolic condition that contributes to increased risk of cardiovascular disease and pancreatitis. The 505(b)(2) regulatory pathway allows Acasti to streamline the overall development program required to support a New Drug Application (NDA) by relying on the safety data of an approved drug.
"We are confident that the results of this study support the 505(b)(2) regulatory pathway chosen by Acasti to gain marketing approval of CaPre," said Jan D'Alvise, president and CEO of Acasti Pharma. "With this momentum, we look forward to working with the FDA to confirm the pathway and optimize the design of our Phase 3 program, which will seek to demonstrate the safety and efficacy of CaPre in patients with severe hypertriglyceridemia."
Acasti's open-label, randomized, four-way, cross-over, bioavailability study compared CaPre given as a single dose of 4 grams in fasting and fed states with the approved hypertriglyceridemia drug LOVAZA (omega-3-acid ethyl esters) in 56 healthy volunteers. The study met its primary objective and demonstrated that the levels of omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) following administration of CaPre did not exceed the levels following administration of LOVAZA in subjects who were fed a high-fat meal. These results support the basis for claiming a comparable safety profile of the two products.
Furthermore, among subjects in the fasting state, CaPre demonstrated better bioavailability than LOVAZA, as measured by blood levels of EPA and DHA. As previously reported, the bioavailability of CaPre is not significantly reduced when taken with a low-fat meal versus a high-fat meal. This could represent a significant clinical advantage for CaPre since the administration with a low-fat meal represents a more attractive regimen for patients with hypertriglyceridemia who follow a restricted diet.
CaPre is a novel composition of omega-3 phospholipids sourced from krill. Its omega-3s, principally EPA and DHA, are naturally either "free" or bound to phospholipids that help them to be better absorbed into the body. This allows for enhanced bioavailability and EPA and DHA blood levels compared to the "esterified" fish-oil omega-3 options such as LOVAZA. CaPre is designed to modulate the major lipids associated with cardio-metabolic disease: in two previously reported Phase 2 clinical trials, CaPre reduced triglyceride levels, lowered non-high density lipoprotein (non-HDL-C, a useful marker of cardiovascular disease), and increased levels of high density lipoprotein (HDL-C, or "good cholesterol") while having a neutral to positive effect on lowering low density lipoprotein (LDL-C, or "bad cholesterol").
"The CaPre bioavailability study has reinforced the compound's unique attributes in comparison with the leading pharmaceutical agent for hypertriglyceridemia," said Roderick Carter, M.D., chairman of Acasti Pharma. "CaPre demonstrates clinically meaningful effects on many key markers of cardio-metabolic health. With high rates of obesity and diabetes fueling the number of patients with elevated triglycerides and cholesterols, CaPre could fill the need for a best-in-class omega-3 medication that addresses the full lipid profiles of these patients."
About CaPre
Sourced from krill oil, CaPre seeks to provide a full scope of health benefits to patients with hypertriglyceridemia, filling a medical need that no other omega-3 treatment option has been able to address. CaPre successfully completed Phase 2 clinical trials for the treatment of hypertriglyceridemia, a very common metabolic condition in which blood levels of triglycerides, a type of lipid, are elevated, posing a risk to cardiovascular health. Severe hypertriglyceridemia, affecting more than 4 million adults in the U.S. is associated with an increased risk of coronary artery disease and pancreatitis and is often caused or exacerbated by uncontrolled diabetes mellitus, obesity and sedentary habits. CaPre is intended to be taken orally once per day in capsule form.
attenti che l'ultima volta che hanno avuto fda ha preso una martellata senza senso
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
2 importanti eventi :
BIO International Convention 2017
•Presenter: Jan D'Alvise, President and Chief Executive Officer
•Date and Time: June 19, 2017 at 4:30 p.m. PT
•Location: San Diego Convention Center, Theater 2
International Academy of Cardiology Annual Scientific Sessions 2017
22nd World Congress on Heart Disease
•Presenter: Laurent Harvey, B. Pharm. M.Sc., Vice President, Clinical and Non-clinical Affairs
•Date and Time: July 14, 2017 at 4:30 p.m. PT
•Location: The Hyatt Regency Vancouver, Hall A
2 nuovi esperti :
Scientific Advisory Board - Barry A. Franklin, Ph.D. and Robert Hegele, M.D., two leading cardiovascular disease experts, were appointed to Acasti's newly-formed Scientific Advisory Board, lending their expertise in scientific matters and the clinical development of CaPre.
pronti per la fase 3 :
Phase 3 Program Plan - Following an end-of-Phase 2 meeting with the U.S. Food and Drug Administration ("FDA"), Acasti finalized the details of its registration program for CaPre® (omega-3 phospholipid). The company plans to conduct two pivotal, double-blind, placebo-controlled studies to evaluate the efficacy and safety of CaPre in a total of approximately 400 patients with severe hypertriglyceridemia (~200 patients per study). The program is designed to satisfy the requirements of the 505(b)(2) regulatory pathway for a new drug application. The company expects to initiate the Phase 3 program on schedule in the second half of 2017
raccolti fondi per fase 3
Financing - On February 21, 2017, Acasti completed a public offering of units, and private placement of convertible debentures and warrants, for aggregate gross proceeds of approximately $7.7 million.
continuano a fare accordi con vari paesi del mondo x avere l'esclusiva di vendita.
LAVAL, QUEBEC--(Marketwired - Apr 24, 2017) - Acasti Pharma Inc. today announced the granting of additional patents by the Taiwanese and Australian patent offices, further expanding the intellectual property position of CaPre.
The granted patents are valid until 2030 and relate to concentrated therapeutic krill oil-based phospholipid omega-3 compositions covering methods for treating or preventing disorders associated with cardiovascular diseases. These patents add to Acasti's growing portfolio of issued patents in the United States, China, Mexico, Saudi Arabia, Panama, and South Africa. Patent applications with similar claim sets are being pursued in many other jurisdictions Worldwide
recenti acquisti insider:
Director Pierre Fitzgibbon bought 15,000 shares last week for a total of 92,000 shares this year.
accordi di produzione su larga scala con azienda francese :
Acasti Pharma and CordenPharma Announce Large Scale Production of CaPre(R) with New Continuous Manufacturing Process
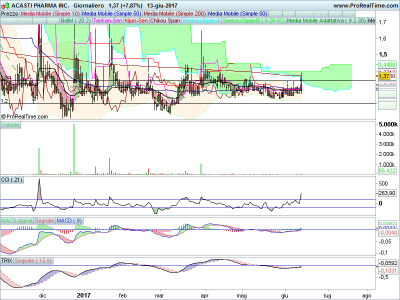
graficamente ieri un primo segnale con un leggero aumento di volumi, tutte le mm in una manciata di centesimi, chiude appena sotto la sma 200 e bordo superiore della cloud,
se buca dovrebbe cominciare un altra storia, io ho un po da recuperare
 errori di gioventù
errori di gioventù 
a qualcuno di voi magari saltan fuori le ferie
BIO International Convention 2017
•Presenter: Jan D'Alvise, President and Chief Executive Officer
•Date and Time: June 19, 2017 at 4:30 p.m. PT
•Location: San Diego Convention Center, Theater 2
International Academy of Cardiology Annual Scientific Sessions 2017
22nd World Congress on Heart Disease
•Presenter: Laurent Harvey, B. Pharm. M.Sc., Vice President, Clinical and Non-clinical Affairs
•Date and Time: July 14, 2017 at 4:30 p.m. PT
•Location: The Hyatt Regency Vancouver, Hall A
2 nuovi esperti :
Scientific Advisory Board - Barry A. Franklin, Ph.D. and Robert Hegele, M.D., two leading cardiovascular disease experts, were appointed to Acasti's newly-formed Scientific Advisory Board, lending their expertise in scientific matters and the clinical development of CaPre.
pronti per la fase 3 :
Phase 3 Program Plan - Following an end-of-Phase 2 meeting with the U.S. Food and Drug Administration ("FDA"), Acasti finalized the details of its registration program for CaPre® (omega-3 phospholipid). The company plans to conduct two pivotal, double-blind, placebo-controlled studies to evaluate the efficacy and safety of CaPre in a total of approximately 400 patients with severe hypertriglyceridemia (~200 patients per study). The program is designed to satisfy the requirements of the 505(b)(2) regulatory pathway for a new drug application. The company expects to initiate the Phase 3 program on schedule in the second half of 2017
raccolti fondi per fase 3
Financing - On February 21, 2017, Acasti completed a public offering of units, and private placement of convertible debentures and warrants, for aggregate gross proceeds of approximately $7.7 million.
continuano a fare accordi con vari paesi del mondo x avere l'esclusiva di vendita.
LAVAL, QUEBEC--(Marketwired - Apr 24, 2017) - Acasti Pharma Inc. today announced the granting of additional patents by the Taiwanese and Australian patent offices, further expanding the intellectual property position of CaPre.
The granted patents are valid until 2030 and relate to concentrated therapeutic krill oil-based phospholipid omega-3 compositions covering methods for treating or preventing disorders associated with cardiovascular diseases. These patents add to Acasti's growing portfolio of issued patents in the United States, China, Mexico, Saudi Arabia, Panama, and South Africa. Patent applications with similar claim sets are being pursued in many other jurisdictions Worldwide
recenti acquisti insider:
Director Pierre Fitzgibbon bought 15,000 shares last week for a total of 92,000 shares this year.
accordi di produzione su larga scala con azienda francese :
Acasti Pharma and CordenPharma Announce Large Scale Production of CaPre(R) with New Continuous Manufacturing Process
graficamente ieri un primo segnale con un leggero aumento di volumi, tutte le mm in una manciata di centesimi, chiude appena sotto la sma 200 e bordo superiore della cloud,
se buca dovrebbe cominciare un altra storia, io ho un po da recuperare

 errori di gioventù
errori di gioventù a qualcuno di voi magari saltan fuori le ferie

Allegati
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
incentivi x fare bene
LAVAL, QUÉBEC, Jun 14, 2017 (Marketwired via COMTEX) -- LAVAL, QUÉBEC--(Marketwired - June 14, 2017) - Acasti Pharma Inc. ("Acasti" or the "Corporation") ACST, +7.22% (ACST), today announced the grant of an aggregate of 1,021,500 stock options to its employees, executives and members of the Board of Directors under the Corporation's Stock Option Plan. Subject to the terms and conditions of the plan, these options will vest in equal annual installments over a period of three years and will entitle the holders to purchase each common share of Acasti at a price of CDN $1.77, until June 14, 2027.
LAVAL, QUÉBEC, Jun 14, 2017 (Marketwired via COMTEX) -- LAVAL, QUÉBEC--(Marketwired - June 14, 2017) - Acasti Pharma Inc. ("Acasti" or the "Corporation") ACST, +7.22% (ACST), today announced the grant of an aggregate of 1,021,500 stock options to its employees, executives and members of the Board of Directors under the Corporation's Stock Option Plan. Subject to the terms and conditions of the plan, these options will vest in equal annual installments over a period of three years and will entitle the holders to purchase each common share of Acasti at a price of CDN $1.77, until June 14, 2027.
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
se buca dovrebbe cominciare un altra storia, io ho un po da recuperare errori di gioventù
a qualcuno di voi magari saltan fuori le ferie
ehehehe due post fa avevo solo sbagliato ferie, erano quelle di natale
io ne ho vendute una parte sopra i 3 e ora sono rientrato....il prossimo giro potrebbe essere ben più consistente, è solo questione di tempo.
p.s. titolo del ....zzo non per deboli di cuore
a qualcuno di voi magari saltan fuori le ferie
ehehehe due post fa avevo solo sbagliato ferie, erano quelle di natale
io ne ho vendute una parte sopra i 3 e ora sono rientrato....il prossimo giro potrebbe essere ben più consistente, è solo questione di tempo.
p.s. titolo del ....zzo non per deboli di cuore
Allegati
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
Acasti Pharma Announces the Closing of Over-Allotment Option for its Public Offering of Common Shares
LAVAL, QUÉBEC--(Marketwired - Jan. 22, 2018) - Acasti Pharma Inc. (NASDAQ:ACST)(TSX VENTURE:ACST), a biopharmaceutical innovator focused on the research, development and commercialization of its prescription drug candidate CaPre® (omega-3 phospholipid) for the treatment of severe hypertriglyceridemia, today announced that the underwriters of its offering of 9,900,990 common shares, together with warrants to purchase up to 8,910,891 common shares (the "Offering") have given notice to exercise their over-allotment option by purchasing an additional 766,179 common shares at a price of US$1.01 per share, for additional gross proceeds to Acasti of approximately US$773,000. Closing of the exercise of the over-allotment option occurred on Monday, January 22, 2018
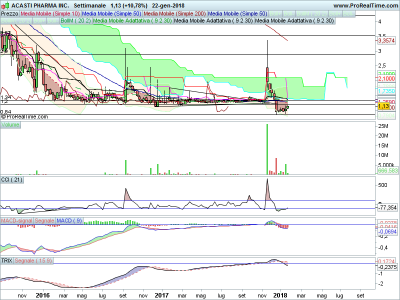
offerta pubblica chiusa, qualche soldino è entrato (10 milioncini), intanto si porta sopra la kijun sen e va a fare una chiusura sui massimi, se buca il muro formato dalle sma 50 e 200 tra 1.31 e 1.34 siamo a cavallo, terzo giorno con volumi in aumento,
primo target 1.80 secondo 2.28
LAVAL, QUÉBEC--(Marketwired - Jan. 22, 2018) - Acasti Pharma Inc. (NASDAQ:ACST)(TSX VENTURE:ACST), a biopharmaceutical innovator focused on the research, development and commercialization of its prescription drug candidate CaPre® (omega-3 phospholipid) for the treatment of severe hypertriglyceridemia, today announced that the underwriters of its offering of 9,900,990 common shares, together with warrants to purchase up to 8,910,891 common shares (the "Offering") have given notice to exercise their over-allotment option by purchasing an additional 766,179 common shares at a price of US$1.01 per share, for additional gross proceeds to Acasti of approximately US$773,000. Closing of the exercise of the over-allotment option occurred on Monday, January 22, 2018
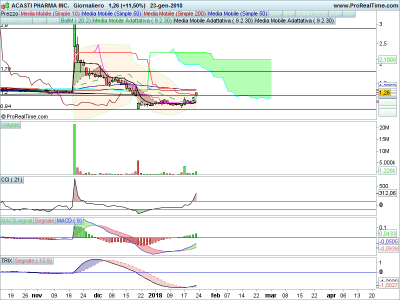
offerta pubblica chiusa, qualche soldino è entrato (10 milioncini), intanto si porta sopra la kijun sen e va a fare una chiusura sui massimi, se buca il muro formato dalle sma 50 e 200 tra 1.31 e 1.34 siamo a cavallo, terzo giorno con volumi in aumento,
primo target 1.80 secondo 2.28
Allegati
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
Acasti Pharma Inc. ( ACST ) ( ACST.V ) (la " Società " o " Acasti ") è lieta di annunciare che ha presentato un prospetto informativo preliminare in forma abbreviata in collegamento con un'offerta pubblica ...............................
simpaticissimi
renna63
Nuovo Utente
- Registrato
- 17/7/09
- Messaggi
- 2.816
- Punti reazioni
- 85
doccia fredda 
LAVAL, Quebec, April 24, 2018 (GLOBE NEWSWIRE) -- Acasti Pharma Inc. (NASDAQ:ACST) (TSXV:ACST) (the “Company” or “Acasti”) is pleased to announce that, in connection with its overnight marketed public offering previously announced on April 23, 2018, it has entered into an underwriting agreement with Mackie Research Capital Corporation (the “Underwriter”) to sell 9,530,000 units of the Company (“Units”) at a price of CDN$1.05 per Unit (the “Offering Price”), with each such Unit comprising one common share (“Common Shares”) and one common share purchase warrant (“Warrants”) of the Company exercisable at any time up to 5 years following the closing of the Offering at an exercise price of CDN$1.31 per Common Share, for aggregate gross proceeds to Acasti of CDN$10,006,500 (the “Offering”).
The Company has also granted the Underwriters an over-allotment option to purchase up to 1,429,500 Units, at the Offering Price, exercisable at any time up to 30 days after the closing date of the Offering, for additional aggregate gross proceeds to Acasti of CDN$1,500,975.
The Offering is being completed at a critical juncture for the Company, as currently more than 50 clinical sites have been initiated and are actively screening and enrolling patients in the two TRILOGY Phase 3 clinical studies for Acasti’s product candidate CaPre® (omega-3 phospholipid). Achievement of this important funding milestone ensures that patient randomization can continue to progress as planned without interruption. This Offering addresses the previously disclosed stated need for the Company to raise additional capital in the current timeframe to support the continued development of CaPre and the ongoing expansion of business development and pre-commercialization activities.
The net proceeds received by the Company from the Offering are more specifically intended to be used by the Company for the further development of CaPre and the continued advancement of the Company’s Phase 3 program, including continued clinical site activation; progression of patient enrollment and randomization; production of additional clinical material (both CaPre and placebo); initial planning of Acasti’s regulatory (NDA) submission; expansion of business development activities; working capital; and other general corporate purposes
The Offering is being made pursuant to a short-form prospectus filed in each of the provinces of Québec, Ontario, Alberta, Manitoba and British Columbia. The Units may also be offered for sale in the United States pursuant to exemptions from the registration requirements of the U.S. Securities Act of 1933. The Units (including the Common Shares and Warrants) will not be registered under the U.S. Securities Act of 1933, as amended, or any state securities laws, and may not be offered or sold in the United States absent registration or an exemption from the registration requirements. This press release shall not constitute an offer to sell or the solicitation of an offer to buy nor shall there be any sale of the Units in any jurisdiction in which such offer, solicitation or sale would be unlawful.
The Offering is scheduled to close on or about the week of May 7, 2018, and is subject to certain conditions including, but not limited to, the receipt of all necessary regulatory approvals including the approval of the TSX Venture Exchange (“TSXV”) and the NASDAQ Stock Market (“NASDAQ”).
LAVAL, Quebec, April 24, 2018 (GLOBE NEWSWIRE) -- Acasti Pharma Inc. (NASDAQ:ACST) (TSXV:ACST) (the “Company” or “Acasti”) is pleased to announce that, in connection with its overnight marketed public offering previously announced on April 23, 2018, it has entered into an underwriting agreement with Mackie Research Capital Corporation (the “Underwriter”) to sell 9,530,000 units of the Company (“Units”) at a price of CDN$1.05 per Unit (the “Offering Price”), with each such Unit comprising one common share (“Common Shares”) and one common share purchase warrant (“Warrants”) of the Company exercisable at any time up to 5 years following the closing of the Offering at an exercise price of CDN$1.31 per Common Share, for aggregate gross proceeds to Acasti of CDN$10,006,500 (the “Offering”).
The Company has also granted the Underwriters an over-allotment option to purchase up to 1,429,500 Units, at the Offering Price, exercisable at any time up to 30 days after the closing date of the Offering, for additional aggregate gross proceeds to Acasti of CDN$1,500,975.
The Offering is being completed at a critical juncture for the Company, as currently more than 50 clinical sites have been initiated and are actively screening and enrolling patients in the two TRILOGY Phase 3 clinical studies for Acasti’s product candidate CaPre® (omega-3 phospholipid). Achievement of this important funding milestone ensures that patient randomization can continue to progress as planned without interruption. This Offering addresses the previously disclosed stated need for the Company to raise additional capital in the current timeframe to support the continued development of CaPre and the ongoing expansion of business development and pre-commercialization activities.
The net proceeds received by the Company from the Offering are more specifically intended to be used by the Company for the further development of CaPre and the continued advancement of the Company’s Phase 3 program, including continued clinical site activation; progression of patient enrollment and randomization; production of additional clinical material (both CaPre and placebo); initial planning of Acasti’s regulatory (NDA) submission; expansion of business development activities; working capital; and other general corporate purposes
The Offering is being made pursuant to a short-form prospectus filed in each of the provinces of Québec, Ontario, Alberta, Manitoba and British Columbia. The Units may also be offered for sale in the United States pursuant to exemptions from the registration requirements of the U.S. Securities Act of 1933. The Units (including the Common Shares and Warrants) will not be registered under the U.S. Securities Act of 1933, as amended, or any state securities laws, and may not be offered or sold in the United States absent registration or an exemption from the registration requirements. This press release shall not constitute an offer to sell or the solicitation of an offer to buy nor shall there be any sale of the Units in any jurisdiction in which such offer, solicitation or sale would be unlawful.
The Offering is scheduled to close on or about the week of May 7, 2018, and is subject to certain conditions including, but not limited to, the receipt of all necessary regulatory approvals including the approval of the TSX Venture Exchange (“TSXV”) and the NASDAQ Stock Market (“NASDAQ”).
doccia fredda
LAVAL, Quebec, April 24, 2018 (GLOBE NEWSWIRE) -- Acasti Pharma Inc. (NASDAQ:ACST) (TSXV:ACST) (the “Company” or “Acasti”) is pleased to announce that, in connection with its overnight marketed public offering previously announced on April 23, 2018, it has entered into an underwriting agreement with Mackie Research Capital Corporation (the “Underwriter”) to sell 9,530,000 units of the Company (“Units”) at a price of CDN$1.05 per Unit (the “Offering Price”), with each such Unit comprising one common share (“Common Shares”) and one common share purchase warrant (“Warrants”) of the Company exercisable at any time up to 5 years following the closing of the Offering at an exercise price of CDN$1.31 per Common Share, for aggregate gross proceeds to Acasti of CDN$10,006,500 (the “Offering”).
The Company has also granted the Underwriters an over-allotment option to purchase up to 1,429,500 Units, at the Offering Price, exercisable at any time up to 30 days after the closing date of the Offering, for additional aggregate gross proceeds to Acasti of CDN$1,500,975.
The Offering is being completed at a critical juncture for the Company, as currently more than 50 clinical sites have been initiated and are actively screening and enrolling patients in the two TRILOGY Phase 3 clinical studies for Acasti’s product candidate CaPre® (omega-3 phospholipid). Achievement of this important funding milestone ensures that patient randomization can continue to progress as planned without interruption. This Offering addresses the previously disclosed stated need for the Company to raise additional capital in the current timeframe to support the continued development of CaPre and the ongoing expansion of business development and pre-commercialization activities.
The net proceeds received by the Company from the Offering are more specifically intended to be used by the Company for the further development of CaPre and the continued advancement of the Company’s Phase 3 program, including continued clinical site activation; progression of patient enrollment and randomization; production of additional clinical material (both CaPre and placebo); initial planning of Acasti’s regulatory (NDA) submission; expansion of business development activities; working capital; and other general corporate purposes
The Offering is being made pursuant to a short-form prospectus filed in each of the provinces of Québec, Ontario, Alberta, Manitoba and British Columbia. The Units may also be offered for sale in the United States pursuant to exemptions from the registration requirements of the U.S. Securities Act of 1933. The Units (including the Common Shares and Warrants) will not be registered under the U.S. Securities Act of 1933, as amended, or any state securities laws, and may not be offered or sold in the United States absent registration or an exemption from the registration requirements. This press release shall not constitute an offer to sell or the solicitation of an offer to buy nor shall there be any sale of the Units in any jurisdiction in which such offer, solicitation or sale would be unlawful.
The Offering is scheduled to close on or about the week of May 7, 2018, and is subject to certain conditions including, but not limited to, the receipt of all necessary regulatory approvals including the approval of the TSX Venture Exchange (“TSXV”) and the NASDAQ Stock Market (“NASDAQ”).
mi son permesso di evidenziare onde non confodere in US come stavo per fare io