Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
CRMD - Cormedix - Ci sfileranno anche questa ?
- Creatore Discussione Gordfin
- Data di inizio
-
 Trading Day 19 aprile Torino - Corso Gratuito sull'investimento
Trading Day 19 aprile Torino - Corso Gratuito sull'investimento

Migliora la tua strategia di trading con le preziose intuizioni dei nostri esperti su oro, materie prime, analisi tecnica, criptovalute e molto altro ancora. Iscriviti subito per partecipare gratuitamente allo Swissquote Trading Day.
Per continuare a leggere visita questo LINK
Più opzioni
Chi ha risposto?Mr China Investiment
Nuovo Utente
- Registrato
- 19/5/00
- Messaggi
- 22.068
- Punti reazioni
- 913
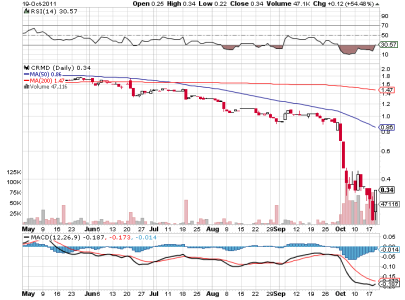
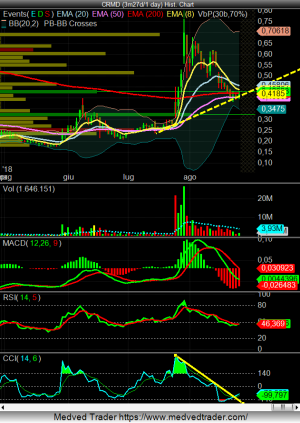
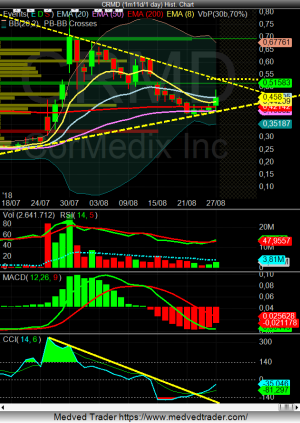
a grafico ha moltissimo spazio target 1.38 sul weekly
volumi eccezionali...
un altra eng???
volumi eccezionali...
un altra eng???
maumal
Super Member
- Registrato
- 22/9/08
- Messaggi
- 2.596
- Punti reazioni
- 57
CorMedix, Inc. Finalizes Pivotal Phase 3 Study Protocol for FDA
BRIDGEWATER, N.J., June 23, 2014 /PRNewswire/ -- CorMedix Inc. (nyse mkt:CRMD), a pharmaceutical company focused on developing and commercializing therapeutic products for the prevention and treatment of cardiorenal and infectious disease, today announced the progress of on-going discussions with the U.S. Food and Drug Administration for a planned pivotal Phase 3 randomized controlled trial ("RCT") for Neutrolin® for use in hemodialysis patients with a central venous catheter. CorMedix worked with the FDA to develop the protocol design for the planned trial for a marketing application. Based on FDA input, the planned Phase 3 clinical trial will be a multi-center, randomized, controlled study conducted in the U.S. and Europe. Dr. Michael Allon, Professor, Department of Medicine, Division of Nephrology, University of Alabama, Birmingham will be the Study Chair of the Neutrolin Phase 3 program.
In addition, CorMedix has earlier submitted detailed information to TUV-SUD for the purpose of advancing our label expansion in the European Union to include oncology, total parenteral nutrition and intensive care patients.
Randy Milby, Chief Executive Officer of CorMedix, stated, "The planned Neutrolin pivotal Phase 3 clinical trial is a major milestone which provides us with a clearly defined development and regulatory pathway for Neutrolin, and we would like to thank the FDA for its invaluable guidance throughout this process. "We believe that Neutrolin is well positioned to unlock significant shareholder value."
About CorMedix Inc.
CorMedix Inc. is a commercial-stage pharmaceutical company that seeks to in-license, develop and commercialize therapeutic products for the prevention and treatment of cardiac, renal and infectious diseases. CorMedix's first commercial product in Germany is Neutrolin®, a catheter lock solution for the prevention of catheter related bloodstream infections and maintenance of catheter patency in tunneled, cuffed, central venous catheters used for vascular access in hemodialysis patients. Please see the company's website at Home - CorMedix Inc. for additional information.

BRIDGEWATER, N.J., June 23, 2014 /PRNewswire/ -- CorMedix Inc. (nyse mkt:CRMD), a pharmaceutical company focused on developing and commercializing therapeutic products for the prevention and treatment of cardiorenal and infectious disease, today announced the progress of on-going discussions with the U.S. Food and Drug Administration for a planned pivotal Phase 3 randomized controlled trial ("RCT") for Neutrolin® for use in hemodialysis patients with a central venous catheter. CorMedix worked with the FDA to develop the protocol design for the planned trial for a marketing application. Based on FDA input, the planned Phase 3 clinical trial will be a multi-center, randomized, controlled study conducted in the U.S. and Europe. Dr. Michael Allon, Professor, Department of Medicine, Division of Nephrology, University of Alabama, Birmingham will be the Study Chair of the Neutrolin Phase 3 program.
In addition, CorMedix has earlier submitted detailed information to TUV-SUD for the purpose of advancing our label expansion in the European Union to include oncology, total parenteral nutrition and intensive care patients.
Randy Milby, Chief Executive Officer of CorMedix, stated, "The planned Neutrolin pivotal Phase 3 clinical trial is a major milestone which provides us with a clearly defined development and regulatory pathway for Neutrolin, and we would like to thank the FDA for its invaluable guidance throughout this process. "We believe that Neutrolin is well positioned to unlock significant shareholder value."
About CorMedix Inc.
CorMedix Inc. is a commercial-stage pharmaceutical company that seeks to in-license, develop and commercialize therapeutic products for the prevention and treatment of cardiac, renal and infectious diseases. CorMedix's first commercial product in Germany is Neutrolin®, a catheter lock solution for the prevention of catheter related bloodstream infections and maintenance of catheter patency in tunneled, cuffed, central venous catheters used for vascular access in hemodialysis patients. Please see the company's website at Home - CorMedix Inc. for additional information.
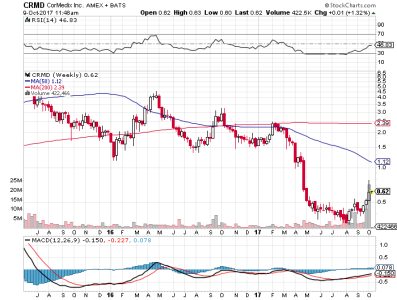
mazza gia passato 1 mese
mcq ridentro ieri e sembrebbe insiemea a qualcun altro $CRMD CorMedix, Inc. stock and investing information on StockTwits (CRMD)
sperem
mcq ridentro ieri e sembrebbe insiemea a qualcun altro $CRMD CorMedix, Inc. stock and investing information on StockTwits (CRMD)
sperem
merlin8121
Nuovo Utente
- Registrato
- 18/2/17
- Messaggi
- 2.437
- Punti reazioni
- 180
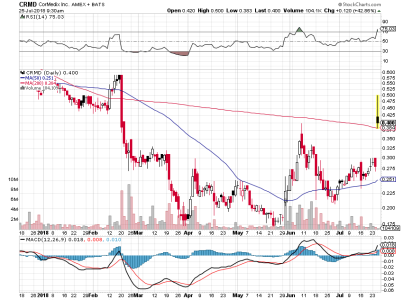
Quella news invece dovrebbe essere molto buona e forse siamo al punto di risalita se tiene la linea.
Praticamente hanno sospeso il trial in anticipo su raccomandazione del comitato indipendente , dal momento che i risultati erano molto buoni. Si dovrebbero riunire con FDA nel 4th trimestre per vedere se FDA accetta direttamente l'NDA. Se cosi fosse farà un altro bel salto, anche più forte che a luglio.
Interessante per il RUNUP sicuramente (se tiene la trendline anche meglio).
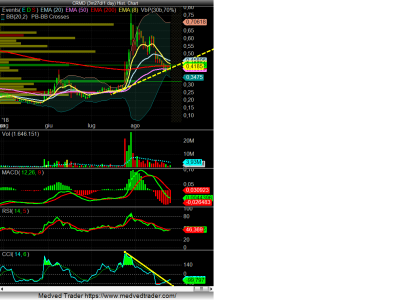
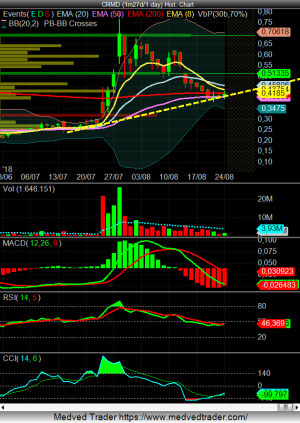
La linea tracciata sul CCI indicherebbe un buy macrocscopico, in base alle strategie di alcuni trader. Poi boh, nulla è assicurato, ma...
Praticamente hanno sospeso il trial in anticipo su raccomandazione del comitato indipendente , dal momento che i risultati erano molto buoni. Si dovrebbero riunire con FDA nel 4th trimestre per vedere se FDA accetta direttamente l'NDA. Se cosi fosse farà un altro bel salto, anche più forte che a luglio.
Interessante per il RUNUP sicuramente (se tiene la trendline anche meglio).

La linea tracciata sul CCI indicherebbe un buy macrocscopico, in base alle strategie di alcuni trader. Poi boh, nulla è assicurato, ma...
Allegati
Ultima modifica:
merlin8121
Nuovo Utente
- Registrato
- 18/2/17
- Messaggi
- 2.437
- Punti reazioni
- 180